DWR-2206
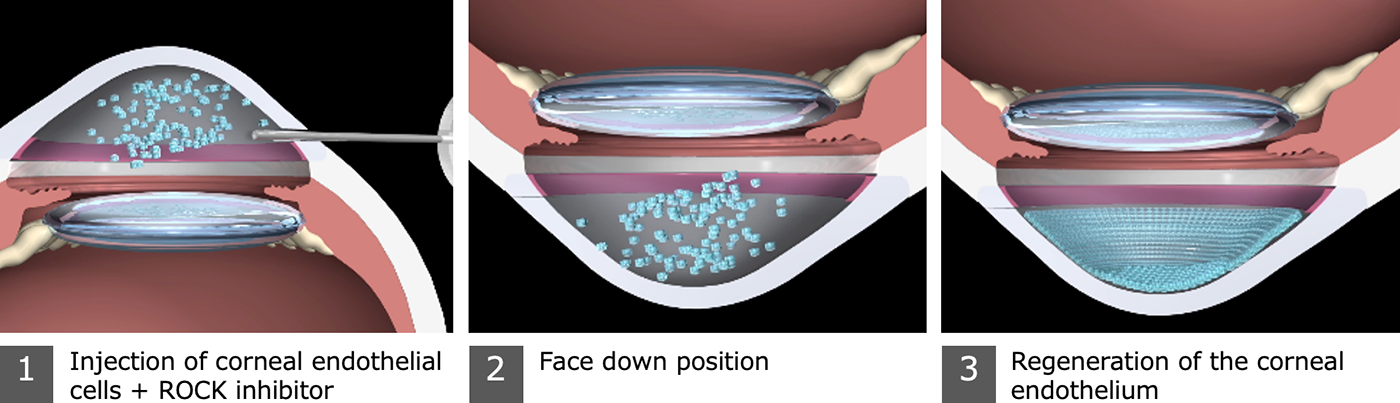
DWR-2206 is a regenerative medicine cell-product for bullous keratopathy. Cultured human corneal endothelial cells of DWR-2206, a suspension containing a ROCK inhibitor, are injected into the anterior chamber to regenerate corneal endothelium. The product has been developing at ActualEyes Inc., a biotech company originating from Doshisha University until now.
Bullous keratopathy is a disease in which corneal endothelial cells are damaged, causing corneal edema, resulting in a cloudy cornea and significant loss of visual acuity. It is caused by a decrease in corneal endothelial cells due to ophthalmic surgery such as Fuchs endothelial corneal dystrophy, cataracts, and glaucoma, and is a progressive condition of corneal endothelial damage. Currently, the only treatment is corneal transplantation, but there are problems such as worldwide shortage of donor corneas and graft failure, and the surgery is considered to be very difficult.
This product is a new treatment that solves these problems and will be developed in Japan in collaboration with ActualEyes Inc. in the future.
- Approach for DWR-2206
 |
||||||
|---|---|---|---|---|---|---|
| ● in Japan | ||||||
| Clinical indication | Bullous keratopathy |
|---|---|
| Development process | Phase II clinical trials (Japan) |
| Collaborative development company | ActualEyes Inc. |
